Light-controlled beta blockers show promise for new and improved medicines
Researchers used X-ray lasers, including SLAC’s LCLS, to control a modified cardiovascular drug with light and captured snapshots showing how it binds to proteins.
By Ula Chrobak
Key takeaways:
- Beta blockers bind to protein receptors that are key to fight-or-flight responses, leading to effects such as lowered heart rate and blood pressure.
- Using X-ray free-electron lasers at SLAC and in Switzerland, an international team of researchers investigated a beta blocker modified with a light-sensitive bond.
- They controlled the drug’s interaction using light and reconstructed X-ray images of the reaction, demonstrating how light could be used to improve medications.
Researchers are illuminating a new route for drug delivery – literally, by controlling drugs with light. Recently, an international team led by the Swiss Paul Scherrer Institute and including researchers from the Department of Energy’s SLAC National Accelerator Laboratory used light to control a modified beta blocker and took X-ray laser snapshots of its interaction with a protein receptor.
Not only did the team demonstrate they could control the beta blocker medicine with light, but they also captured 3D images of the interaction at multiple time points. The images revealed that light can switch the beta blocker between different positions on the receptor, which suggests it may be possible to fine tune the drug’s potency while it’s in the body. The findings, published in the journal Angewandte Chemie, also demonstrate how X-ray lasers like the Linac Coherent Light Source (LCLS) can be harnessed to study medicines at the atomic level. This can aid the design of drugs that precisely target protein receptors and therefore have fewer side effects.
“There are significant medical implications,” said Sandra Mous, a coauthor and staff scientist at SLAC. “This is one of the first demonstrations of how we can use LCLS and these light-triggered compounds to get a better understanding of protein receptor dynamics.”
Light-powered molecular switches
In the new study, the team focused on adrenergic receptors. These proteins bond with the hormones epinephrine and norepinephrine to produce fight-or-flight responses via increases in blood flow and heart rate. Drugs called beta blockers bind to the same locations on the receptor proteins, which stops the hormones from latching on and in turn reduces heart rate and blood pressure. This can benefit patients with heart conditions.
In previous work, collaborators at the Institute for Advanced Chemistry of Catalonia modified a common beta blocker, propranolol, by adding a light-activated bond to the molecule. For this study, the team created crystals of the molecule and the protein receptor. In experiments conducted at LCLS and the Swiss X-ray Free-Electron Laser, they fed these crystals into the instruments. First, they zapped the crystal with light, toggling the molecular switch. Then, at two different time increments afterward, the researchers shot X-ray pulses at samples, capturing the diffraction pattern of the rays in a detector.
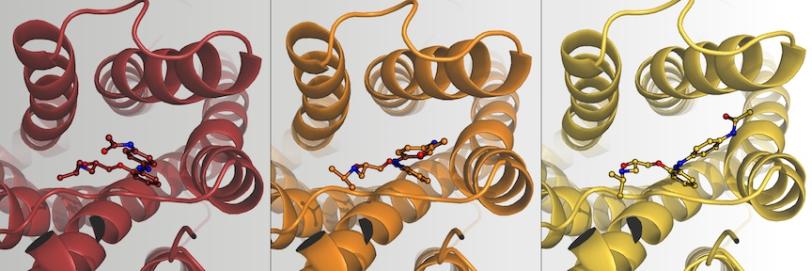
The team used the diffraction data to reconstruct three snapshots of the reaction: the dark phase (before light activation), intermediate phase (17 nanoseconds after light activation), and the light phase (10 seconds after light activation). In the images, they observed that in the dark phase, the beta blocker fits snugly in a pocket of the receptor; in this position, it strongly blocks the activity of the protein receptor. At 17 nanoseconds after being excited with light, it wiggles around in the pocket. After 10 seconds, it had scooted between two helical strands of the receptor, assuming a new, twisted position that was still attached to the receptor.
“We were super surprised that it stays in the binding pocket,” said Joerg Standfuss, the paper’s senior author and a professor at the Paul Scherrer Institute in Switzerland and the University of Zurich. The researchers had expected that light would fully detach the beta blocker, turning the receptor “on.”
X-rays for drug discovery
“This new binding mode is unlike any other drug that’s been observed,” said Robin Stipp, the study’s lead author and now a postdoctoral researcher at Uppsala University in Sweden. When the molecule is in the light-activated position, the receptor is active but appears that it would bind hormones less efficiently. The findings suggest it may be possible to adjust the activity of the beta blocker up and down using light, which could have potential benefits such as more precise medicine delivery and limiting side effects.
The finding also shows it may be possible to use light-controlled drugs to regulate the activity of the broader family of adrenergic receptors called G protein-coupled receptors, which are all similar in structure. These receptors are virtually everywhere in our bodies and a major drug target. “They are involved in many sensing and signaling pathways,” said Stipp. “Now, we have maybe taken the first steps to modulating their activity.”
While light-activated drugs are still a long way from the pharmacy shelf, the study suggests new avenues for fine-tuning how medicines interact with protein receptors. The 3D images the team reconstructed also show how a standard analogy used in biochemistry falls short. Often, signaling molecules are described as “keys” that fit in the “lock” of corresponding receptors, switching their activity off and on. But, in real life, “it’s always a dynamic model,” said Standfuss, where the molecule influences the receptor pocket and vice versa. Using light controls and ultrafast X-ray lasers, “we are actually starting to see these concepts experimentally,” said Standfuss.
The authors also include researchers at LeadXpro AG, Institute for Advanced Chemistry of Catalonia and the Stanford University School of Medicine. The work was supported by the DOE’s Office of Science and the National Institutes of Health. LCLS is an Office of Science user facility.
Citation: R. Stipp et al., Angewandte Chemie International Edition, 18 March 2026 (https://doi.org/10.1002/anie.202517995)
For media inquiries, please contact media@slac.stanford.edu. For other questions or comments, contact SLAC Strategic Communications & External Affairs at communications@slac.stanford.edu.
About SLAC
SLAC National Accelerator Laboratory explores how the universe works at the biggest, smallest and fastest scales and invents powerful tools used by researchers around the globe. As world leaders in ultrafast science and bold explorers of the physics of the universe, we forge new ground in understanding our origins and building a healthier and more sustainable future. Our discovery and innovation help develop new materials and chemical processes and open unprecedented views of the cosmos and life’s most delicate machinery. Building on more than 60 years of visionary research, we help shape the future by advancing areas such as quantum technology, scientific computing and the development of next-generation accelerators.
SLAC is operated by Stanford University for the U.S. Department of Energy’s Office of Science. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time.